INSTITUTE FOR BIOFUNCTIONAL POLYMER MATERIALS
Hemocompatibility of biomaterials
Core Research


We work to decipher the relevance of material surface properties for the activation of host defence reactions and develop adaptive inhibitor release systems to control these processes.

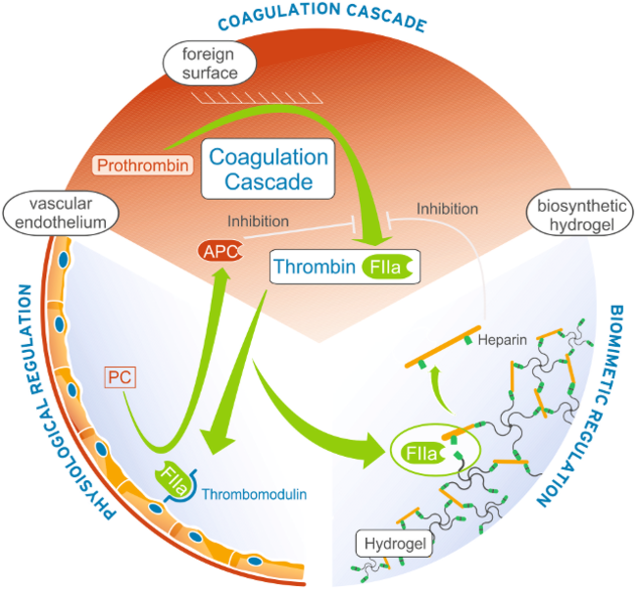
MATERIAL-INDUCED BLOOD ACTIVATION
When blood contacts material surfaces, enzymes of the intrinsic pathway of the coagulation cascade and activating mediators from adherent platelets can induce blood coagulation. We investigate protein adsorption processes which are relevant for the activation of the blood coagulation cascade (FXIIa, FSAP), platelets (fibrinogen) and the immune system (complement cascade). To identify surface characteristics of least activating, most tolerable, hemocompatible materials, our approach is the incubation of material surfaces with fresh whole human blood in customized incubation chambers. This setup provides a realistic blood-material interface with the possibility to vary interaction time, shear forces and blood preconditioning. Blood and surfaces are analyzed for specific markers of interest concerning several blood activation processes.
Selected References
Schulz S., Maitz M. F., Hänsel S., Renner L. D., Werner C. Analyzing the antiseptic capacity of silver-functionalized poly(ethylene glycol)-heparin hydrogels after human whold blood exposure, Biomaterials Science 6, 1129-1139 (2018).
Sperling C., Maitz M. F., Grasso S., Werner C., Kanse S. M. A positively charged surface triggers coagulation activation through factor VII activating protease (FSAP), ACS Applied Materials & Interfaces 9(46), 40107-40116 (2017).
Sperling C., Fischer M., Maitz M. F., Werner C. Neutrophil extracellular trap formation upon exposure of hydrophobic materials to human whole blood causes thrombogenic reactions, Biomaterials Science 5(10), 1998-2008 (2017).
Herklotz M., Hanke J., Hänsel S., Drichel J., Marx M., Maitz M. F., Werner, C. Biomaterials trigger endothelial cell activation in co-incubation with human whole blood, Journal of Biomedical Materials Research Part B: Applied Biomaterials 104, 258-268 (2016).
Hemocompatible coatings
In vivo, coagulation and inflammation are subtly balanced physiological systems of activators and inhibitors. Medical devices in blood often disturb this balance. We develop feedback controlled release systems, which integrate into the physiological control loops. For example, coagulation responsive release systems for heparin were shown to afford outstanding thromboprotection. The concept of adaptive release systems for host response inhibitors is currently extended also to other functions, such as immunomodulation.
Selected References
Johnbosco C., Zschoche S., Nitschke M., Hahn D., Werner, C., Maitz M. F. Bioresponsive starPEG-heparin hydrogel coatings on vascular stents for enhanced hemocompatibility. Materials Science and Engineering: C (128): 112268 10.1038/ncomms3168 (2021).
Maitz M. F., Zitzmann J., Hanke J., Renneberg C., Tsurkan M.V., Sperling C., Freudenberg U., Werner C. Adaptive release of heparin from anticoagulant hydrogels triggered by different blood coagulation factors. Biomaterials 135: 53-61 (2017).